Translational Award To Support Cancer Immunotherapy Technology
Two Johns Hopkins research teams have received technology development grants totaling approximately $200,000 through the Louis B. Thalheimer Fund for Translational Research. One of those teams includes researchers from the Institute for NanoBioTechnology (INBT)
Finalists pitched their proposals virtually in late May to an outside panel of independent researchers and investors, innovation executives and venture investors. Established through a generous $5.4 million gift from businessman and philanthropist Louis B. Thalheimer, the fund provides seed funding for vital proof-of-concept and validation studies of Johns Hopkins technologies.
Since 2016, the Thalheimer Fund has awarded more than $1.7 million to 20 projects at Johns Hopkins. Grants range from $25,000 to $100,000, and all recipients have formally reported their inventions to JHTV. Previous Thalheimer winners are developing a faster and more accurate way to diagnose epilepsy; an oral therapy for patients suffering from inflammatory bowel disease; and a longer-lasting treatment for wrinkles and migraines, among other technologies.

Jonathan Schneck
The team from the INBT includes principal investigator Jonathan Schneck, professor of pathology and affiliate researcher at the INBT, and Hai-Quan Mao, director and core researcher at the INBT and professor of materials science and engineering.
The pitch:
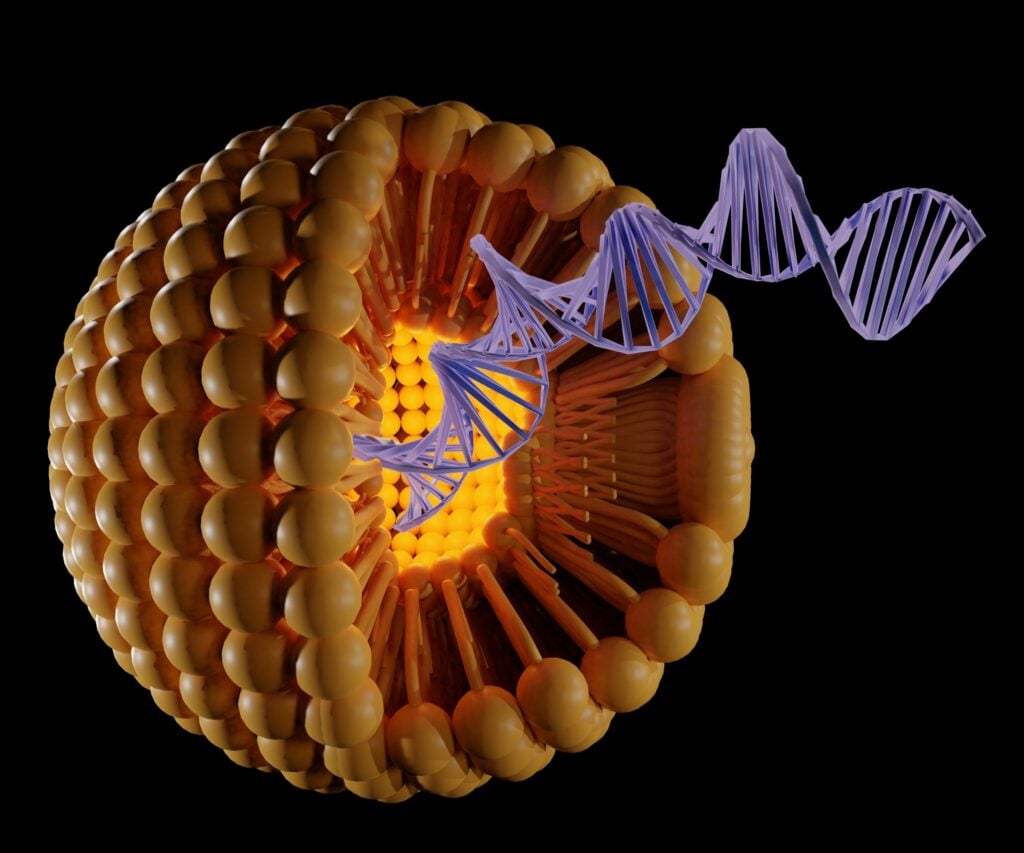
Lipid Nanoparticles Targeting Tumor-Specific CD4+ T-Cells for In Situ CAR T-Cell Generation for Immunotherapy
Jonathan Schneck’s research focuses on the basic mechanisms that control immune responses. Specifically, he studies how cytotoxic T-cells, known as killer T-cells — cells that help eliminate infections or cancers — get activated, recognize their targets and get turned off again. His team focuses on how to manipulate the immune system with the hopes of designing improved anti-cancer therapies by ramping up the immune system or treating autoimmune disease and transplant patients with better immune system suppression.

Hai-Quan Mao
Chimeric antigen receptor (CAR) T-cells developed in labs have demonstrated remarkable clinical efficacy in the blood-born cancers leukemia, lymphoma and multiple myeloma, with long-term remission and even cure being observed.
There are currently six Food and Drug Administration-approved CAR therapies, all utilizing T-cells to target B-cell lymphoma or multiple myeloma. Despite the remarkable impact of these engineered cell products, they are still hindered by barriers that limit their use as mainstream cancer therapies, specifically the lengthy and costly ex vivo cell production, tumor antigen escape, and having little to no efficacy in solid tumors.
Furthermore, manipulating antigen-specific-T cells in vivo is a significant challenge due to the lack of appropriate targeting and efficient delivery platforms.
Schneck and Hai-Quan Mao, along with Ph.D. candidates Joseph Choy and Yining Zhu, have proposed to use a novel nanoparticle platform for highly specific mRNA delivery to T-cells — jointly developed in their laboratories to overcome these barriers — and to create an efficacious therapy for solid tumors. The platform will selectively expand tumor specific CD4+ T-cells while simultaneously endowing them with a CAR in vivo, enabling dual recognition of cancer cell antigens while minimizing deleterious side effects. As a nonviral T-cell transfection approach, the platform itself represents a major advance and can herald new ways of genetically manipulating T-cells in addition to endowing them with CAR T-cell qualities.
While the proposed study will focus on CD4+ T-cells, this platform is versatile and can be tailored to target, transfect, and expand antigen specific CD8+ T-cells as well.
If successful, this disruptive technology has the potential to revolutionize cancer immunotherapies as an off-the-shelf therapeutic modality and overcome the cost, safety and efficacy barriers of existing CAR T-cell therapies.
See the full announcement by Johns Hopkins Technology Ventures for more information.
Latest Posts
-
 Studying Cells to Better Understand Aging
May 29, 2026
Studying Cells to Better Understand Aging
May 29, 2026
-
 Reza Kalhor Receives $250,000 President’s Innovation Award
May 1, 2026
Reza Kalhor Receives $250,000 President’s Innovation Award
May 1, 2026
-
 Alum Receives Schmidt Science Fellowship
April 17, 2026
Alum Receives Schmidt Science Fellowship
April 17, 2026


